Prevention is better than cure

The need to reduce the use of antibiotics in food producing animals requires additional preventive measures for intestinal health. Bacillus sp. PB6 has proven to be an efficient tool to support that. The combination with novel Bacillus spp. FXA and G3 goes further in driving intestinal resilience.
Part of the Gut Health 2022 Special
In animal health management there used to be little specific attention to healthy animals unless it was desirable to vaccinate. If animals still became ill, antibiotics were the treatment of choice. Since the ban of the last AGP (antibiotic growth promoters) in Europe and the dramatic increase of NAE (No Antibiotics Ever) in the US, there has been a rapid global trend to decrease the use of all antibiotics in poultry and livestock production. A recent example in the European Union is the new veterinary regulation 2019/6, which was implemented in January 2022 to further restrict the use of antibiotics in food producing animals.
Therefore, both research and commercial production are strongly pursuing options to manage health preventively by increasing resilience to health challenges. The following article shows one example each from commercial turkey production and from university research in broiler chickens.
Commercial production with Bacillus sp. PB6
In cooperation with a commercial turkey integrator, Bacillus sp. PB6 (Clostat) was applied across all flocks via water (3×108 CFU/L) in instances where indications of beginning intestinal health problems were observed. Data were collected for the first 27 calendar weeks of the year and historic data of the 3 previous years were the negative control. Data collected included diseases, antibiotic treatments and weather information. In the trial period, Bacillus sp. PB6 treatments were added to the recorded parameters.
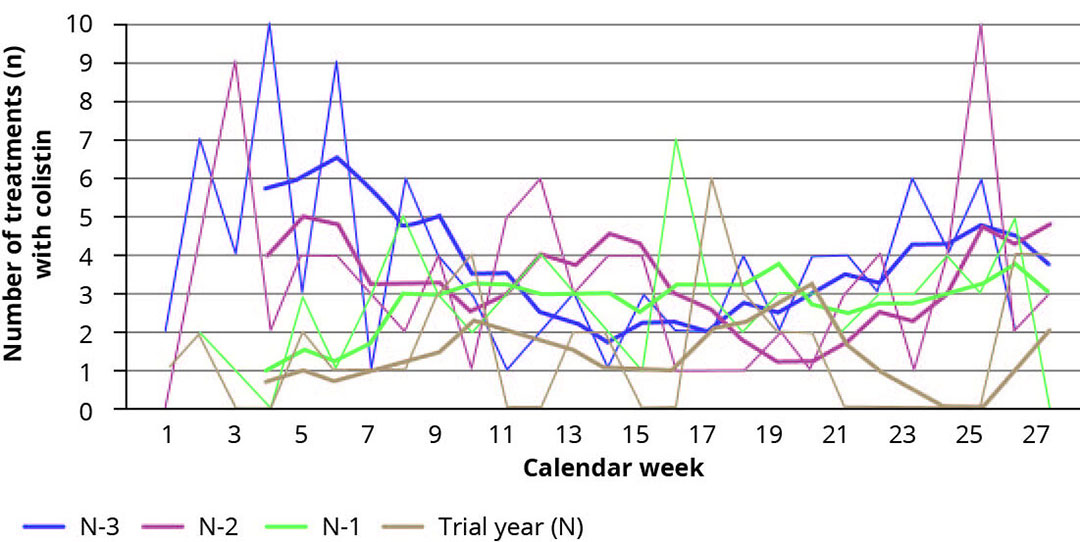
Figure 1 – Number of colistin treatments by weeks.
Effects on antibiotic usage
Figure 1 shows the number of colistin treatments, per week. The usage of colistin was significantly (p-value = 0.004) decreased by 44%. This did not lead to a shift towards other antibiotics, as their use reduced by 51% over the reference period. The effect of Bacillus sp. PB6 application on the usage of beta-lactams was not statistically significant (p< 0.05), there was a numerical decrease of 13% when the usage of beta-lactams in the trial year was compared to the usage in prior year (data not shown).
Disease incidence requiring treatment was recorded by week. The incidence of enteritis reduced by 38%, colibacillosis by 34% and ORT by 38%.
The aim of this trial was to generate hard data that a specific strain of Bacillus sp. (PB6) may reduce therapeutic antibiotic use in commercial farming, which was conclusively demonstrated by this trial.
The reduction in antibiotic treatments did not lead to a deterioration in animal health, but an improvement. The effect on the “other” antibiotics recorded was significantly (p-value 0.002) reduced. Applications of tylosin, doxycycline, fluoroquinolones, TMPs reduced by 51% over the reference period. Colistin is typically used for intestinal disorders. Despite the fact that a greater reduction could be observed in colistin usage, compared to the effect on beta-lactams. Despite this decrease in colistin use, colibacillosis incidence decreased by 34%.
Overall, the trial presented here clearly indicated that it is possible to use a probiotic (Bacillus sp. PB6) to reduce therapeutic antibiotic use in turkeys.
Research with Bacillus sp. PB6, novel Bacillus spp FXA and G3
A new probiotic consisting of multiple health promoting Bacillus spp. strains was formulated based on a previous extensive in vitro and in vivo screening process focusing on anti-Enterobacteriaceae activity, to combine with the proven anti-clostridial activity from Bacillus sp. PB6. In total, 100 one-day-old Ross-308 chicks were divided over 4 groups housed in isolators. Each group of birds received a different diet: a multi-strain probiotic (Enterosure – Bacillus spp. PB6, FXA, G3) dose 1: 3×108 CFU/kg feed, dose 2: 3×109 CFU/kg feed; not supplemented, challenged control (positive control), not supplemented, not challenged control (negative control). At 14, 16 and 18 days of age, birds in all groups except the negative control were infected with 4.5-5.7×108 CFU avian pathogenic E. coli. At 21, 28 and 35 days of life, 5 birds from each group were killed for necropsy (Figure 2).
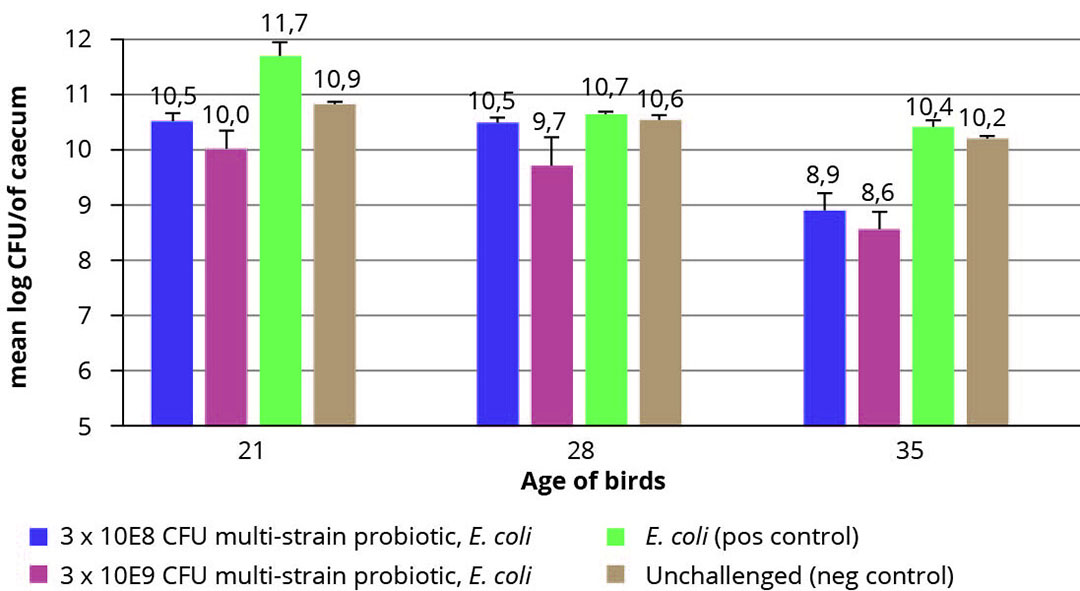
Figure 2 – Mean caecal CFU counts after 3, 4 and 5 weeks by treatment.
Decrease in caecal E. coli
Growth performance differences were observed, with the highest body weight gain and lowest FCR during the last week of the trial noted for the new multi-strain probiotic.
Furthermore, a significant decrease in caecal E. coli counts, compared to the infected control could be observed in the groups treated with both dosages of the multi-strain probiotic 35 days of age, and in addition for the highest dosage at day 21 and 28 (p< 0.05, Figure 3).
This research trial confirmed the potential of the multi-strain probiotic Enterosure to reduce E. coli in poultry. Further work showed efficacy in naturally acquired E. coli challenges in commercial farms.
Conclusion
In conclusion, both the antibiotic reduction by application of Bacillus sp. PB6, and the effect being more pronounced with antibiotics aimed at intestinal disorders could be proven in the commercial field trial. To achieve further reductions in the need to treat with antibiotics, a new idea was therefore to look for more specific action on Enterobacteriaceae. The subsequent screening and university trial confirmed that the effect observed for the single strain Bacillus sp. PB6 could be improved by the addition of 2 additional Bacillus spp. strains, to strengthen efficiency against Enterobacteriaceae of concern (i.e. E. coli).
*Enterosure is not yet available in some geographic regions including the European Union. Certain statements may not be applicable in all geographic regions.
References available on request.






