Understanding inulin and oligofructose in piglet diets
In the post-antibiotic era, it is important to have a complete understanding of how gut bacteria grow and interact with each other, and with the animal. Today, we place significant value on establishing beneficial microflora right after weaning, to the point that such micro-organisms are often provided as in-feed supplements (probiotics). Fully understanding the mechanisms of proper nourishment for such beneficial bacteria remains the ‘frontier’ in piglet nutrition.
By Karel Thurman, Beneo Animal Nutrition, Tienen, Belgium
Oligofructose is a widely known oligosaccharide of fructose that has for quite some time been available as a source of prebiotic fibre in piglet diets. Nevertheless, its use remains the exclusive privilege of those who have followed the development of prebiotic fibres and appreciate the relationship between a healthy gut microflora and piglet growth performance. Essential to this understanding is the understated need of proper ‘nourishment’ for the beneficial gut microbes. And, in essence oligofructose is just that – food for beneficial gut bacteria.
To understand the role of oligofructose in developing a healthy gut system, we need to follow its path down the gastrointestinal tract from the moment it enters the stomach. Before we begin this journey, it is beneficial to know more about the origin and structure of this bacterial ‘food’. One of the major and most widely researched oligofructose is inulin. It is abundant in root plants such as artichokes, chicory, onions, leeks, and asparagus and is a blend of short- and medium-length chains of fructose molecules. These chains are non-digestible by pigs due to the way the fructose molecules are linked to each other by a special beta-2-1-glycosidic bond. This specific bond gives inulin all its properties as a prebiotic fibre. When inulin is enzymatically hydrolysed, the average chain length is reduced and we get a similar blend but rich in oligosaccharides, with slightly different chemical properties (but similar characteristics).
Chicory is the richest source of inulin in nature (around 16% inulin) and the primary source of commercial inulin extracts. Crystalline inulin is slightly sweet and highly soluble in warm water, which makes it an ideal ingredient for certain types of milk replacers.
Starting in the stomach
Fructans play a beneficial role at different levels of the digestive tract, to start with the stomach. One of the main functions of the stomach is as a barrier against the entrance of pathogenic bacteria; a function achieved through the secretion of hydrochloric acid that creates a very acidic (unfavourable) environment for most pathogens. In piglets, however, this mechanism is not yet fully developed, with ensuing diarrhoea being the most common observation. In suckling pigs, milk provides lactose that apart from feeding the piglet also feeds lactic bacteria. These bacteria produce lactic acid to replace the function of hydrochloric acid. However in weaned pigs, where milk products are (due to the price) included in restricted amounts in the diets, feed intake is rather limited. As a result the lactose reaching the stomach is often not sufficient to provide nourishment for lactic bacteria, which leads to a less acidic environment. Inulin and oligofructose stimulate the growth of bifidobacteria and lactic acid bacteria which in turn produce volatile fatty acids (VFA) which contribute to the pH barrier and limit pathogen growth. In that sense commercial lactose replacement products often include inulin as it helps to account for the pH decreasing effect of lactose.
In the small intestine
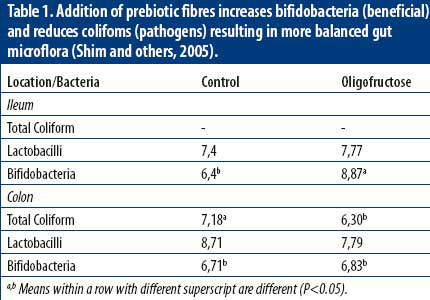
Upon leaving the stomach, we encounter an almost pH-neutral environment, which is regulated by the animal with outmost rigidity. Here, it is almost impossible to lower the acidity of the gut contents in order to harm pathogens. In contrast, another mechanism comes into play that limits the chances of survival for pathogenic bacteria: competitive exclusion. In simple words, all bacteria have to compete with each other for available nutrients, while at the same time secreting their own ‘antibiotics’ to eliminate competitors. When for example beneficial bacteria, like lactic and bifidobacteria, outgrow the most common pathogens (coliforms and salmonellae) there is a greatly diminished chance that these opportunistic pathogenic bacteria become a real problem. So, piglets should be provided a source of prebiotic fibre (that preferentially feeds beneficial bacteria), to hinder the growth of pathogens which favour the rather high level of protein in common piglet diets (Table 1). This explains why piglets often suffer from diarrhoea.
In the large intestine
Fermentation, that is uptake of nutrients by beneficial bacteria for use as fuel for their own needs, yields not only lactic acid, as is the case with lactic bacteria, but also a number of volatile fatty acids (VFA). In a recent study (Xu and others, 2005), it was demonstrated that oligofructose supplementation increased total VFA production from 83 mmol/g digesta to 108 mmol/g, an increase of 30%. These VFA have a double role. First, they provide a small amount of energy to the animal as they are being absorbed by the intestinal epithelium. Although this amount of energy accounts for less than 5% of total energy needs, it is important to note that young piglets are always in an energydeficient state, and as such, each small contribution is significant. The second and most important role of VFA is of course as a deterrent against pathogenic bacteria. In fact, such acids as acetic acid and propionic acid are often used as feed ingredients for the same purpose. In addition a certain VFA, butyric acid, plays a key role in stimulating the regeneration process in damaged epithelial tissue. This of course is of tremendous importance as it is well known that recently weaned pigs suffer from damaged gut infrastructure due to starvation, bacterial toxins, and feed allergens. It is interesting to consider that although butyric acid remains a feed ingredient sold at a premium price, provision of inulin, and oligofructose in general, can provide such acid at the level required and at a fraction of the cost.
Other effects
From a review of available research reports, a number of other beneficial ‘side effects’ can be cited that only add to the value of inulin and oligofructose. These include:
- Regulation and balance of gut micro flora population, which is of benefit as any such upsets always lead to digestive problems, ending with diarrhoea.
- Stimulation of the immune system, most likely through the effect of interaction between beneficial bacteria and the host (animal).
- Increased calcium absorption (oligofructose enriched inulin supports the absorption of more essential minerals from food). This effect is not of insignificant value in pig nutrition, as it can have a place in low-calcium piglet feeds (calcium reduces stomach acidity), and also in diets for sows that frequently suffer from leg (bone) problems ending in premature culling.
- Regulation of bowel movements, that prevent both constipation (a problem in sows) and soft faeces (a non-pathogenic but undesirable problem in piglets receiving highdensity diets).
- Ammonia emission reduction as the pH reduction shifts bacterial metabolism from proteins to carbohydrates, yielding a cleaner house environment and more sustainable livestock production systems.
Growth and feed intake
Naturally, inulin and oligofructose help to maintain good gut health. However, it is widely appreciated that gut health and high post-weaning performance are closely related. Such notion is supported by numerous trials as well as by a recent study which also demonstrated a reduction in diarrhoea and mortality (Table 2).
From the above it can be concluded that prebiotic fibres can be of tremendous help in maintaining gut health. To this end, examples of inclusion levels in practical pig diets are presented in Table 3.
Join 18,000+ subscribers
Subscribe to our newsletter to stay updated about all the need-to-know content in the pigsector, three times a week. Beheer
Beheer










 WP Admin
WP Admin  Bewerk bericht
Bewerk bericht